Therapeutics
Latest News
Latest Videos

CME Content
More News
China clears first mite-targeting drop for Demodex blepharitis, bringing XDEMVY-style relief and new options beyond lid scrubs and tea tree oil.


Previously, the company had received CRLs from the FDA in April 2025 and November 2023 for NDAs for reproxalap for the treatment of dry eye.
University of Utah data show US ophthalmic drug and eye drop shortages last longer and occur more often, affecting local and systemic medications equally.
PULSAR post-hoc finds aflibercept 8 mg matches 2 mg for PCV, offering 12–16-week dosing and reduced injection burden.

Explore latest RVO care: faricimab, Eylea HD, and biosimilars that cut injection burden and protect vision from macular edema.

FDA clears DifGen’s generic fluorometholone 0.1% eye drop suspension, expanding lower-cost options for anterior eye inflammation and steroid care.

Most patients do well on topical therapy, but innovation in IOP-lowering drops remains essential.

Lab tests show red‑light myopia devices exceed ANSI safety limits in seconds, fueling urgent calls for independent validation before kids use them.

Tenpoint Therapeutics’ YUVEZZI, expected in the US in Q2 2026, is the first fixed-dose combination drop designed to improve near vision via pupil modulation with once-daily dosing.
FDA authorizes compassionate use of urcosimod for neuropathic corneal pain, a condition that causes severe pain and sensitivity of the eyes, face, or head.

According to the company, the FDA’s decision was supported by visual function results from the phase 2 ACUITY trial in ON.

Jennifer E. Thorne, MD, PhD, discusses evidence from the ADJUST trial, including relapse risk, retreatment success, and how clinicians should monitor children when considering adalimumab discontinuation.
Strategies for treatment intervals, drug selection, and patient expectations.

From artificial intelligence to home monitoring, Joel Schuman, MD, of Wills Eye Hospital, explores the innovations that could change how clinicians detect and treat glaucoma in the new year.

Panel examines drug mechanisms, patient factors, and strategies to optimize results.


Formosa Pharmaceuticals partners with Rxilient Medical to commercialize APP13007, a new ophthalmic treatment for post-surgery inflammation and pain relief.


The acquisition includes product candidates MELT-210, MELT-300, and MELT-400 based on the Zydis ODT (oral dissolving tablet) drug delivery platform.
Ophthalmologists weigh in on evolving diagnostic tools, emerging therapies, and practical approaches for managing today’s most challenging ocular-surface cases.
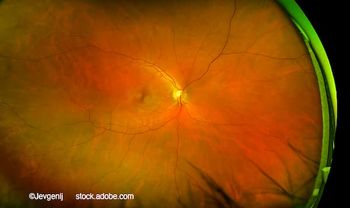
Harpal Sandhu, MD, FRCSC, discusses the preclinical performance of XPK-640 and how Optigo’s hyaluronic acid-anchoring platform may reshape dosing expectations in retinal disease.
The results presented at 2025 European Society of Medical Oncology meeting support neoadjuvant darovasertib as a potential treatment to delay or prevent progression of the disease.

Emerging anti-VEGF agents offer enhanced durability and anatomic outcomes in retinal disease.
Chiu discusses factors affecting adoption of the FDA-approved epi-on therapy, from clinic setup to insurance coverage, while highlighting its safety and efficacy.