Conference Coverage
26 days ago
ARVO 2026: Global RETFound studyLatest News

Self-Screening for Ocular Surface Malignancies Using a Smartphone

Everest Medicines acquires Greater China rights to Aceclidine Presbyopia Eye Drop (VIZZ)

Closing the gap: John Berdahl, MD, on the global cataract burden and the emerging dementia connection

FDA guidance to help accelerate cell and gene therapies

The Residency Report: Cardiovascular risk scores as a window into ocular disease

Shorts










Ophthalmology Times Digital Edition




Podcasts
Continuing Medical Education
All News

The company confirmed it does not plan to submit a New Drug Application (NDA) to the US Food and Drug Administration (FDA)

The topical synthetic peptide receives orphan drug and fast track status and enters phase 2 as a first-in-class treatment for a rare degenerative corneal disease.

Samsung Bioepis launched Opuviz, an aflibercept biosimilar, in Europe for wet AMD, DME, retinal vein occlusion, and myopic CNV.

The study, led by researchers at Keio University School of Medicine, measured near add power in 6258 patients aged 40 to 79.

Andrew G. Lee, MD, and Drew Carey, MD, discuss recurrence of papilledema after discontinuation of GLP-1 receptor agonists and the implications for monitoring patients with idiopathic intracranial hypertension.
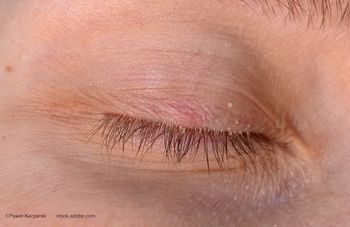
Two cases illustrate how recognizing and treating Demodex can change surgical outcomes and resolve chronic dry eye disease.

A pilot study shows that analyzing how the cornea moves under air-puff stimulation may improve early keratoconus detection.

The resubmission follows a successful appeal by the company with the Office of New Drugs (OND).

Knowing pregnancy-related risks enhances patient safety.

Therini Bio begins a phase 1b DME trial of THN391, an intravitreal antibody targeting fibrin-driven retinal inflammation.

The next-generation FSYX ocular pressure adjusting pump adds wireless connectivity to transmit patient usage data to prescribing practices.
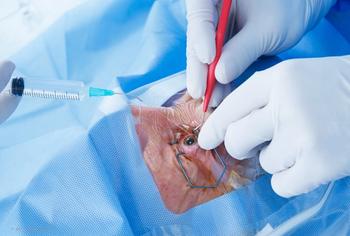
Off-label use of laboratory-grade dye preparation found to harbor Sarocladium kiliense beyond the sterilizing capacity of standard filtration.

Learning Brazilian Portuguese boosts brain resilience and may delay dementia symptoms, but a humorous lesson highlights the realities of health care paperwork and waiting rooms.

The device, described in a new study, uses a technique called temporal interference-based transcorneal electrical stimulation, or TI-TES.

The FDA's OND found substantial evidence of effectiveness for bevacizumab-vikg in nAMD, clearing the way for a BLA resubmission in June 2026 and a decision expected within 60 days.