
Efficacy of treatment option is also confirmed in pediatric population in India.

Efficacy of treatment option is also confirmed in pediatric population in India.
The Lasker-DeBakey Clinical Medical Research Award, dubbed ‘America’s Nobel,’ recognizes the wide use of optical coherence tomography to manage eye disease, prevent blindness.

The company announced its previously announced agreement to divest its late-stage ophthalmic assets to Laboratoires Théa S.A.S. has been terminated.

Ocuphire Pharma and Viatris developed the drug together for the reversal of pharmacologically-induced mydriasis (RM) produced by adrenergic agonist or parasympatholytic agents.
Preservative-free Iyuzeh, launched more than 10 years ago, currently is available in 46 countries, mostly under the brand name Monoprost, with about 1.5 million patients treated monthly, according to the company.

Discover how advocacy can impact your clinical work and ultimately patient care.
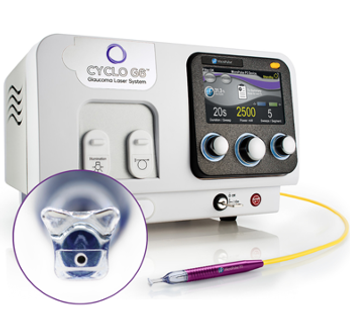
Highlighting the benefits of Iridex's updated P3 Probe and Cyclo G6® Laser software for MicroPulse® transscleral laser therapy. The redesigned probe improves ergonomics and visibility, while the user-friendly software ensures precise, reproducible glaucoma treatments.

Study finds 45% of injuries involve the eyes.

Peter J. McDonnell, MD, and Neda Shamie, MD, discuss the transformative impact of minimally invasive glaucoma surgery (MIGS) on glaucoma care and its integration with cataract surgery, sharing insights and practical tips for ophthalmologists.

Procedure corrects small-angle strabismus and removes need for prism glasses

On the 'Negotiations' episode of RWO: The Podcast, Dr. Lisa Nijm joins Drs. Nicole Bajic and Grayson Armstrong to discuss personal experiences with contract negotiation.
According to a news release, the J-code for SYFOVRE will become effective on October 1, 2023.
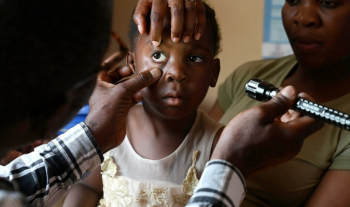
The Orbis Flying Eye Hospital will return to Zambia for a 3-week training project in Lusaka coinciding with World Sight Day on October 12.
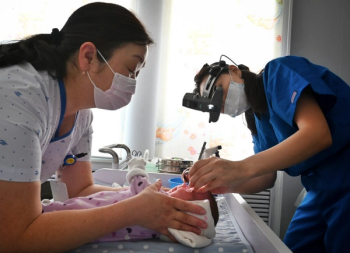
According to the companies, the partnership will roll out a proprietary system that has earned FDA breakthrough designation, one of the few ophthalmic devices to achieve this status in almost a decade.


News
To educate patients on the disease, the company has enlisted the help of actor Eric Stonestreet and his mother, Jamey.

Corneal wound burn is a critical, yet often overlooked, complication in cataract surgery. Recognizing early signs is crucial.
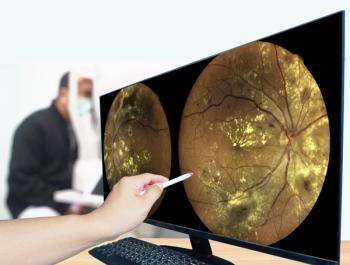
Kiora CEO Brian Strem, PhD, offers an update on the development of KIO-301, a molecule that selectively targets retinal ganglion cells, giving them the ability to sense light.
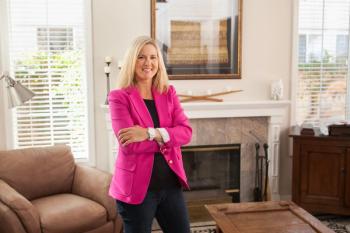
As a gold medalist with profound vision loss, she will lead advocacy efforts for people with glaucoma that are designed to empower patients to participate actively in their glaucoma treatment journey

Enhance your ophthalmology skills with expert tips from RWO podcast. Match procedures to patients, seek guidance, review surgical notes, practice patience, and be honest with patients.


The research will focus on the compound's impact on biomolecular condensates, which are implicated as a key driver of pathology in neurodegeneration and diseases impacting high metabolic organs. Because the retina is one of the highest energy consuming systems in the human body, deficits in energy supply can be catastrophic.

Clinicians must screen, diagnose, and manage for effective treatment.

Researchers from the University of Barcelona are looking to unlock the function of genes in order to design therapies to treat patients diagnosed with retinitis pigmentosa.

Balancing motherhood & ophthalmology: Prioritize family planning, set boundaries, integrate work & personal life. Insights from ophthalmologist-moms.

Resolution of macular leakage is an early biomarker of vascular stability.


An overview of Jesse Berry’s advice for starting and growing a social media account

Andrew Lee, MD, and Andrew Carey, MD, sit down on another episode of the NeuroOp Guru to discuss visual outcomes in solid versus cystic pituitary adenomas.