Refractive Surgery
Latest News

Latest Videos

CME Content
More News

The acquisition also includes the ALLY Robotic Cataract Laser Treatment System, LENSAR’s proprietary Streamline software technology and LENSAR legacy laser system.

Maria Vittoria Cicinelli, MD, Denise Loya-Garcia, MD, PhD, and Vishal Raval, MD, were announced as the recipients and will receive a $3,000 honorarium.
The device offers a shortened procedure time and efficient, effective SMILE for patients with myopia, with or without astigmatism.

With this addition, Zeiss completes the Corneal Refractive Workflow, which already includes Visumax 800 and SMILE pro.
Deepinder Dhaliwal, MD, LAc, explores advancements in managing Fuchs endothelial corneal dystrophy, highlighting Descemet stripping only (DSO). Long-term outcomes show promising results, supporting DSO as a viable alternative to traditional donor-tissue-based keratoplasty procedures.

Connect, learn, and innovate in a family-friendly atmosphere: What to expect at EnVision Summit 2025
The EnVision Summit, co-founded by Bonnie An Henderson, MD, is a unique family-oriented, multispecialty ophthalmology CME conference, along with a newly introduced optometry track. Henderson sits down with Ophthalmology Times to talk about the 2025 conference that will take place February 14 to 17 at the Caribe Hilton San Juan, in Puerto Rico.

A time line marked by innovation, collaboration, and improving patient outcomes.

The DOG recognised Heidelberg Engineering founder Christoph Schoess, Prof Herbert Kaufmann, and Prof Ioannis G. Pallikaris

In a study, a team of Chinese researchers looked at the mechanisms behind the development of EI and explored effective treatment options.


The Summit will feature 18+ course hours and an extensive resource library to support practice adoption of AT IOLs

The LYNX-2 Phase 3 study is evaluating phentolamine ophthalmic solution 0.75% for the treatment of decreased visual acuity under low light conditions following keratorefractive surgery

The device is intended for use for LASIK vision correction surgery for myopia and myopic astigmatism.

According to the company, Phase 3 startup activities are underway, and the clinical trial is expected to start in the first quarter of 2024.

Procedure can reduce scleral biomechanical stiffness and rejuvenate dynamic range of focus.

Light-adjustable lens in patients with previous RK: Improved visual and refractive outcomes after cataract surgery.

Clear communication, transparent priceing, options are key to positive experience

Language can drive understanding, trust, and better outcomes for patients

New laser is designed to deliver low-energy treatment with ultrashort pulse duration.

Procedure offers sharp contrast to cutting procedures without risk, pain, or drama.
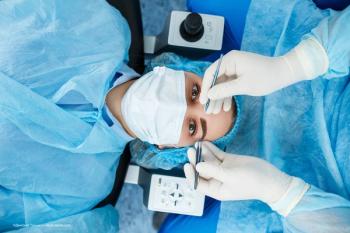
ICLs offer a strong surgical correction option for some patients.
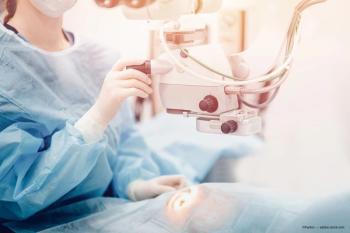
In a presentation at the EnVision Summit 2023 in Puerto Rico, Sumayya Ahmad, MD, pointed out that for patients with lower Schirmer’s scores and corneal staining, PRK may be a good choice. If the dry eye can be treated preoperatively and the eye has no staining, surgeons can consider LASIK.

Mark H. Blecher, MD, dives into his AAO 2022 topic: "One-Year Visual Outcomes Following Monocular Implantation with a Small Aperture IOL."
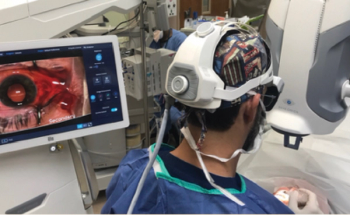
Physicians can use navigational tools to attain a much more immersive perspective.

Catching up after ASCRS 2022, Ora's Caitlin Black, senior director and therapeutic area head of medical devices, talks about the next generation of ophthalmic device therapies and diagnostics, clinical trial updates and new innovations that are most exciting for presbyopia treatment.