
Patients who underwent LASIK had undergone phacoemulsification 7 years earlier compared to a matched control group, according to the study authors.

Patients who underwent LASIK had undergone phacoemulsification 7 years earlier compared to a matched control group, according to the study authors.

Even small changes can have a large impact, saving time for physicians.

Study demonstrates that patients achieved long-term visual acuity gains following CureSight treatment.

OCU410 is a modifier gene therapy candidate being developed for geographic atrophy, an advanced stage of dry age related macular degeneration.

Scientists at the Doheny Eye Institute, in collaboration with UCLA chemists, demonstrate how a single gene mutation in an enzyme complex known to produce the energy required by cells causes a sudden, blinding disease.
The transient receptor potential melastatin 8 (TRPM8) are cold-sensitive thermoreceptors that play central role in tear film homeostasis

The Clareon platform will be available in India in 2 Presbyopia correcting IOL technologies: the Clareon PanOptix and Clareon Vivity.

The collaboration combines Clario's transformative technology with Cleveland Clinic's market-leading imaging expertise and AI-enabled analysis to help bring new therapies to patients, faster.

Ophthalmologists can educate their patients about the importance of using certified solar viewing glasses or other safe viewing methods.

Vermont is the latest state to consider legislation expanding the scope of practice for optometrists. Earlier this year, South Dakota became the 12th state to expand the scope of practice for optometrists.

The organization aims to educate viewers of dangers and safety precautions ahead of the eclipses.

LL-BMT1 is a novel, preservative-free, weekly drug-eluting contact lens created by the company’s proprietary 3D printing technology.

Cyclosporine superior to vehicle, improves corneal staining in dry eye disease

Delivered in a water-free solution, Vevye’s new formulation offers a high concentration of cyclosporine that works rapidly, has no preservatives, and rarely causes irritation

Investigators emphasized that the data does not indicate a causal relationship between eye disorder and systemic diseases.

In the study, published in Nature Biotechnology, researchers used their new system to correct disease-causing mutations in the eyes of two mouse models of genetic blindness, partially restoring their vision.

Their machine-learning multistage system predicts the expected disease progression of patients and their VA with age-related macular degeneration (AMD), diabetic macular edema (DME), and retinal vein occlusion (RVO).


The study showed that a 12-day treatment of KIO-101 topically at multiple doses was well tolerated in healthy volunteers and patients with inflammation of the eye.

Results show Eylea HD extended dosing regimens were non-inferior to aflibercept 2 mg injection (Eylea) for both the treatment of wet age-related macular degeneration (wAMD) and diabetic macular edema (DME).

TL-925 is a first in class, topical Bruton’s tyrosine kinase (BTK) inhibitor that prevents mast cell activation and degranulation, and also blocks the cytokine-driven inflammatory response, and restores lacrimal unit homeostasis, offering the potential for disease modification
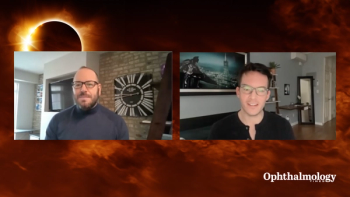
Jeff Todd, president and CEO of Prevent Blindness, and astrophotographer Jon Carmichael sat down with David Hutton of Ophthalmology Times to talk about how ophthalmologists can help make sure patients are viewing the eclipse safely as well as proper ways to view/photograph the event without damaging your eyes or equipment.

According to the company, the milestone underscores Vivity’s wavefront-shaping technology, which simplifies presbyopia correction for surgeons and patients.
Study concluded that the short-term results of LBN to treat mild-to-modern OAG and OHT was safe and efficacious.

The company has divested its Coherus Ophthalmology subsidiary to Sandoz, Inc. for upfront all-cash consideration of $170 million.
News
Eyenovia acquired the US commercial rights for the drug last August from Formosa Pharmaceuticals.

Investigators believe that AMD and cancer may have similar risk factors in common.

ChatGPT-4 (OpenAI) had an overall “fair” performance when answering multiple-choice ophthalmic questions unrelated to multifocal imaging.

Andrew G. Iwach, MD, recounts two personal life experiences that underscore the significance of patient-centered care in glaucoma management.

Early detection of the disease is key to optimal patient outcomes