
News
Researchers examined disparities in service coverage of pediatric ophthalmologists and pediatric optometrists in relation to patient demographics to cast a light on at-risk populations and provide data for future decisions.

News
Researchers examined disparities in service coverage of pediatric ophthalmologists and pediatric optometrists in relation to patient demographics to cast a light on at-risk populations and provide data for future decisions.

The tool shows disease progression and vision loss simulations for multiple diseases such as AMD, XLRP, and achromatopsia

David Hutton, Managing Editor of Ophthalmology Times sat down with Claris Bio CEO, Clarke Atwell, to discuss the company's path toward developing treatment for neurotrophic keratitis.

Donahue will serve as president until June 30, 2025, when he will host the AAPOS 2025 annual meeting in Salt Lake City, Utah. He previously had served as vice president since July 2023.

The company expects to launch the supplement in the US, which features a proprietary blend of ingredients including lutein, zeaxanthin isomers, curcumin and vitamin D3, under the brand name Blink NutriTears early in the third quarter of 2024.

Akari Therapeutics, Ocuphire Pharma, and PharmAbcine are among the companies that have shared their plans for presentations at the ARVO annual conference in Seattle, Washington.

The foundation noted the summit will be held June 25-27 in Mexico City, Mexico, and will feature stakeholders across the eye health landscape with a common goal of eradicating avoidable blindness by 2030.
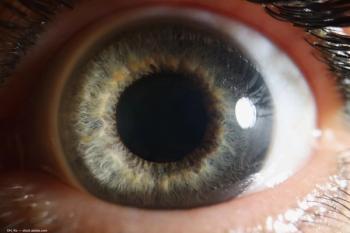
The company announced results from a Phase IV study of tropicamide and phenylephrine hydrochloride ophthalmic spray 1%/2.5% (Mydcombi) designed to characterize the efficacy and duration of the lowest deliverable dose.

The module encompasses real world data on more than 250,000 de-identified patients and provides information on extracting diagnoses from clinical notes in electronic health records.

CRISPR manipulation presents opportunities and challenges.

At this year's ASCRS meeting in Boston, Massachusetts, Shoham-Hazon, MD provided highlights from his two paper presentations.

The collaboration will bring automated, AI-powered OCT segmentation to MICRON Software Suite.

The WVU Health System Board of Directors recently approved $233.5 million to fund the construction of a multi-center outpatient facility with additional exam and testing rooms.

Cataract simulation platform uses virtual reality to train ophthalmologists

At the 2024 ASCRS meeting, David M. Lubeck detailed highlights from his presentations on the minimally invasive glaucoma surgery (MIGS) space.
According to the company, OCU410 utilizes an AAV delivery platform for the retinal delivery of the RORA gene.

Annual conference to kick off May 5 in Emerald City.
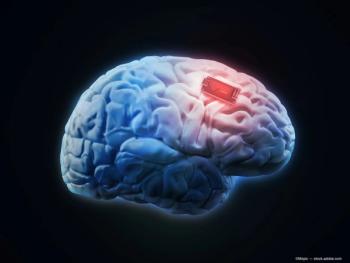
The system is comprised of 25 miniaturized wireless stimulators with 400 electrodes that are implanted permanently.

At this year's ASCRS meeting in Boston, Massachusetts, Josefina Botta, MD, MSc, discussed her real-world experiences with the RayOne EMV toric lens.

At this year's ASCRS meeting Rom Kandavel, MD, of Colvard-Kandavel Eye Center in Los Angeles, California discussed the paper he presented at the meeting, which focused on IOP lowering and medication burden for patients who received the iStent inject.

SGT-1001 uses its novel gene coding technology for gene integration via transposition. It plans to advance SGT-1001 into the clinic in the first half of 2025.

Shift could lead to changes in the current management of retinal diseases
News
According to the company, 46.2% of patients demonstrated a 1- or 2-step improvement in the Diabetic Retinopathy Severity Scale (DRSS) at 40 weeks in the Axpaxli arm, compared to 0% in the control arm.

BVI lenses feature a unique C-loop haptic design, known as “POD,” introduced to the market with the launch of the PODEYE hydrophobic IOL in Europe in 2012.

Analysis showed that patients with dry eye had “worse sleep quality than the healthy population."

Powers, whose father, sister, and brother are ophthalmologists, has held senior executive positions in strategic planning, marketing, and commercial and medical affairs at vision-focused companies.
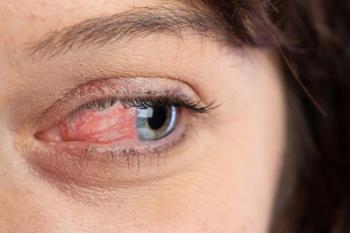
Look beyond isolated treatment of the eye to address systemic factors.

Adam Muzychuk, MD, BSc, FRCSC discusses his presentation, "Outcomes of an ultra-low cylinder powered (0.90) toric versus non-toric in low astigmatic patients undergoing cataract surgery."

A brief analysis showed that ChatGPT 3.5 performed better.

According to the company, the feedback from the interactive meetings will provide guidance for the path to a PMA submission.