
NYU Langone’s Jordan Mandell, MD, and Ariana Levin, MD, sit down with UCLA’s Anne Coleman, MD, to discuss her recent study on cardiovascular risk scores and their association with glaucoma, AMD, and other ocular diseases.

NYU Langone’s Jordan Mandell, MD, and Ariana Levin, MD, sit down with UCLA’s Anne Coleman, MD, to discuss her recent study on cardiovascular risk scores and their association with glaucoma, AMD, and other ocular diseases.

The company confirmed it does not plan to submit a New Drug Application (NDA) to the US Food and Drug Administration (FDA)

The topical synthetic peptide receives orphan drug and fast track status and enters phase 2 as a first-in-class treatment for a rare degenerative corneal disease.

Samsung Bioepis launched Opuviz, an aflibercept biosimilar, in Europe for wet AMD, DME, retinal vein occlusion, and myopic CNV.

The study, led by researchers at Keio University School of Medicine, measured near add power in 6258 patients aged 40 to 79.

Andrew G. Lee, MD, and Drew Carey, MD, discuss recurrence of papilledema after discontinuation of GLP-1 receptor agonists and the implications for monitoring patients with idiopathic intracranial hypertension.
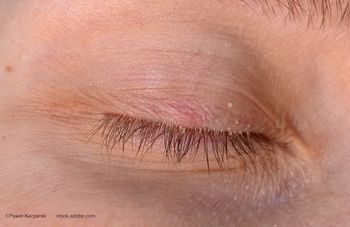
Two cases illustrate how recognizing and treating Demodex can change surgical outcomes and resolve chronic dry eye disease.

A pilot study shows that analyzing how the cornea moves under air-puff stimulation may improve early keratoconus detection.

The resubmission follows a successful appeal by the company with the Office of New Drugs (OND).

Knowing pregnancy-related risks enhances patient safety.

Therini Bio begins a phase 1b DME trial of THN391, an intravitreal antibody targeting fibrin-driven retinal inflammation.

The next-generation FSYX ocular pressure adjusting pump adds wireless connectivity to transmit patient usage data to prescribing practices.
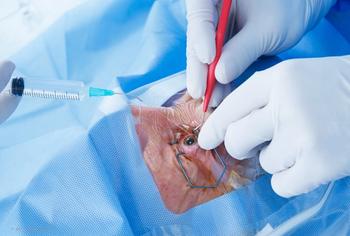
Off-label use of laboratory-grade dye preparation found to harbor Sarocladium kiliense beyond the sterilizing capacity of standard filtration.

Learning Brazilian Portuguese boosts brain resilience and may delay dementia symptoms, but a humorous lesson highlights the realities of health care paperwork and waiting rooms.

The device, described in a new study, uses a technique called temporal interference-based transcorneal electrical stimulation, or TI-TES.

The FDA's OND found substantial evidence of effectiveness for bevacizumab-vikg in nAMD, clearing the way for a BLA resubmission in June 2026 and a decision expected within 60 days.

NUS researchers report that plant-based photosynthetic nanoparticles restored corneal NADPH levels and reversed damage within 5 days in preclinical dry eye.

Bausch + Lomb's PreserVision AREDS3 combines the established AREDS2 micronutrient formula with a B-vitamin complex.

Michigan pediatric ophthalmologist advances childhood glaucoma care with longer-lasting Ahmed valves, Ologen, and new clues on prematurity glaucoma.

Transient vision loss may foreshadow stroke or heart events; study urges urgent 90-day evaluation and long-term cardiovascular monitoring.

Three cases illustrate how earlier, proactive intervention can reduce medication burden and preserve vision across a range of glaucoma severity.
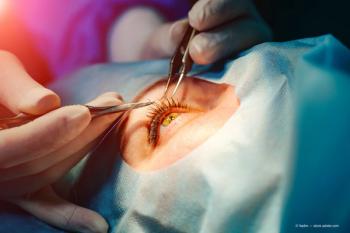
Two years after LASIK, hidden corneal nerve loss and immune activation persist, with higher IL‑16 and faster T‑cell motion despite normal exams.

Swiss regulator backs tinlarebant for Stargardt as DRAGON phase 3 shows slower retinal atrophy, bringing first treatment closer to patients.

Investigators noted the patient's symptoms "reliably abated when she played the oboe or when she sang, only to return immediately after stopping.”

The app is designed to improve confidence among neurology and ophthalmology trainees.

Sunil S. Patel, MD, PhD, of the University of Texas Southwestern Medical Center, breaks down QA102's first-in-class mechanism targeting harmful gut bacteria and retinal inflammation, what the phase 2 drusen volume and GA growth rate data reveal about the therapy's potential, and what a phase 3 program must address to bring an oral AMD treatment to patients without options.

Biogen buys Apellis, taking over Syfovre for geographic atrophy—what it means for GA care, trial data, safety signals, and future formulations.

Ultra-widefield pediatric retinal scans plus adaptable AI reveal hidden eye issues during routine visits, enabling comfortable, scalable screening and earlier specialist referrals.
Danish JAMA Dermatology trials show short-term topical or oral corticosteroids don’t raise intraocular pressure, easing glaucoma worries in atopic dermatitis care.
New Turkish study links sleeve gastrectomy weight loss to clearer cornea and lens, lower eye pressure, and widened anterior chamber in 6 months.