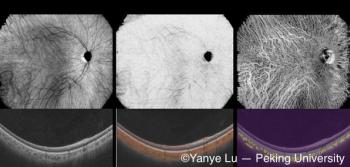
Early results suggest participant diversity and novel measures will enable new, artificial intelligence-driven insights. As the study evolves, it could lead to significant advancements in understanding how environmental exposures contribute not only to diabetes but also to its complications, including those affecting eye health, such as diabetic retinopathy.