New molecule for allergic conjunctivitis makes debut
First new therapy in nearly a decade offers another option for physicians, patients
Special to Ophthalmology Times®
The FDA recently approved the first prescription-only topical ocular formulation of the second-generation antihistamine cetirizine for the treatment of ocular itching associated with allergic conjunctivitis.
Cetirizine ophthalmic solution 0.24% (Zerviate, Nicox Ophthalmics Inc; licensed to Eyevance Pharmaceuticals, LLC) demonstrated robust efficacy in 3 randomized, double-masked, placebo-controlled clinical trials using the Ora Conjunctival Allergen Challenge (CAC) model among patients with allergic conjunctivitis.
Two of the trials that evaluated onset and duration showed that cetirizine ophthalmic solution led to statistically and clinically significantly less ocular itching versus vehicle at 15 minutes and 8 hours after treatment.1
Related:
The ophthalmic solution breaks the 10-year drought since the last approval for allergic conjunctivitis treatment, with the twice-daily drops hitting the market in March 2020.
Cetirizine hydrochloride (Zyrtec, Johnson & Johnson Consumer) is recognized as the number 1 oral antihistamine allergy treatment recommended by allergists, with 23 years on the market and countless doctor and patient years of experience.2
Based on its vast track record of therapeutic success and safety in different formulations, the industry sought to develop cetirizine as an ophthalmic solution.
Background
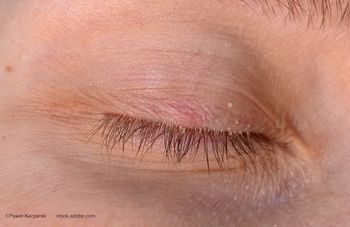
Allergic conjunctivitis affects at least 30% of Americans.3 Reactions range anywhere from mild—making it merely a self-limiting nuisance—to the other end of the spectrum, when allergies become a debilitating disease, causing patients to have a significantly impaired quality of life.
Stimuli, whether tree and grass pollens, animal hair and dander, or any number of other environmental insults, manifest in the familiar cascade of ocular symptoms that include itching, redness, chemosis, tearing, and eyelid swelling.
Related:
Thanks to the vast work being done around
Allergic conjunctivitis is a frequent and substantial piece of the puzzle when it comes to finding the root cause of ocular surface disease.
Patients with allergies typically have bilateral signs and symptoms, with the most common ocular symptoms being itching, burning, redness, and tearing.
Allergic conjunctivitis is often associated with swelling, an important area of differentiation from
Related:
Allergy cycle
In response to an allergen, the process of conjunctivitis has an early acute phase followed by a late phase. Allergens interact with immunoglobulin E, which is bound to sensitized mast cells that in turn activate increased histamine and subsequent degranulation.4
The release of histamine and other proallergic mediators during the acute phase induces itching, vasodilation, and vascular leakage. This is followed by ocular redness, chemosis, and lid swelling.
Mast cells then synthesize, releasing cytokines, chemokines, and growth factors, which kicks off a cascade of inflammatory events.
During the final late-phase reaction, eosinophils, neutrophils, and macrophages infiltrate conjunctival tissues.5,6
The commonly used and approved treatments for
Related:
Ocular form of cetirizine
Cetirizine is a second-generation antihistamine (highly selective H1 receptor antagonist) that binds competitively to histamine receptor sites to reduce swelling, itching, and vasodilation. Two phase 3 efficacy studies revealed strong and similar anti-itch efficacy of cetirizine ophthalmic solution 0.24% compared with vehicle (using the CAC model).
The single-center (study 1) and multi-center (study 2), double-masked, randomized, vehicle-controlled, parallel group CAC studies were conducted over approximately 5 weeks and 4 study visits.
Patients with moderate and severe symptoms were enrolled in the trials, and study 2 required patients to have more severe allergic conjunctivitis symptoms. Subjects were screened for an allergen response at visits 1 and 2 and then randomized at visit 3.
Approximately 100 subjects were randomly assigned in each study. The primary efficacy end points were ocular itching and conjunctival redness 15 minutes and 8 hours posttreatment, post-CAC.
Related:
Quells itching fast
Investigators administered cetirizine 15 minutes or 8 hours before CAC, and subjects had significantly lower ocular itching at all time points post-CAC (P < .0001) compared to vehicle in both studies.
The investigators’ assessment of conjunctival redness was significantly lower after cetirizine treatment compared to vehicle at 7 minutes post-CAC and at both 15 minutes and 8 hours posttreatment in both studies (P < .05).
Numerous secondary end points, ocular and nasal, were also examined.
It should be noted that the most robust treatment differences were observed in study 2, where patients were required to have more severe symptoms in order to be included (P < .05). Investigators identified no safety concerns for cetirizine ophthalmic solution 0.24%.
Related: PODCAST:
Comfort is key
For added comfort, cetirizine ophthalmic solution 0.24% is designed with Hydrella, which includes glycerin and hydroxypropyl methylcellulose, ingredients commonly found in lubricant drops.
Patients in the FDA trials reported a mean comfort score of less than 1 at all time points (on a scale of 1-10, with 1 being the most comfortable). The solution is also formulated with a neutral pH of 7.0, similar to the natural tear film.
Conclusion
Eye-care providers now have a solution for patients with allergic conjunctivitis experiencing disruptive ocular itching.
The drop form of cetirizine ophthalmic solution 0.24% gives eye care providers a chance to greatly improve patients’ quality of life with an effective and targeted approach.
Related:
Notably, unlike some oral antihistamines, the new formulation did not cause drowsiness.
Cetirizine ophthalmic solution 0.24% is a strong therapeutic with an ability to quickly quell ocular itching for patients experiencing allergic conjunctivitis.
There are no safety concerns identified with cetirizine treatment. Drop comfort is rated as “very comfortable.”
About the authors
Richard D. Najac, MD, FACS
p: 718/261-2666
Richard D Najac, MD, FACS, is a second-generation ophthalmologist practicing in Queens, New York, for more than 20 years. Dr Najac is an investor of Eyevance.
Richard Teofilo Atallah
p: 732/445-4636
Richard Teofilo Atallah, PGY-3, is a third-year medical student at Rutgers New Jersey Medical School in Newark, New Jersey,, and has no financial disclosures related to this product.
---
References
1. Meier EJ, Torkildsen GL, Gomes PJ, Jasek MC. Phase III trials examining the efficacy of cetirizine ophthalmic solution 0.24% compared to vehicle for the treatment of allergic conjunctivitis in the Conjunctival Allergen Challenge model. Clin Ophthalmol. 2018;12:2617-2628. doi:10.2147/OPTH.S185835
2. Zyrtec the #1 allergist recommended brand. Johnson & Johnson Consumer Companies, Inc. Accessed May 21, 2020. http://www.multivu.com/ assets/52078/documents/Zyrtec-fact-sheet-99-original.pdf
3. Bielory L. Allergic and immunologic disorders of the eye. Part II: ocular allergy. J Allergy Clin Immunol. 2000;106(6):1019-1032. doi:10.1067/mai.2000.111238
4. La Rosa M, Lionetti E, Reibaldi M, et al. Allergic conjunctivitis: a comprehensive review of the literature. Ital J Pediatr. 2013;39:18. doi:10.1186/1824-7288-39-18
5. Bacon AS, Ahluwalia P, Irani AM, et al. Tear and conjunctival changes during the allergen-induced early- and late-phase responses. J Allergy Clin Immunol. 2000;106(5):948-954. doi:10.1067/mai.2000.110930
6. Ciprandi G, Buscaglia S, Pesce GP, Bagnasco M, Canonica GW. Ocular challenge and hyperresponsiveness to histamine in patients with allergic conjunctivitis. J Allergy Clin Immunol. 1993;91(6):1227-1230. doi:10.1016/0091-6749(93)90327-c
7. Gomes PJ. Trends in prevalence and treatment of ocular allergy. Curr Opin Allergy Clin Immunol. 2014;14(5):451-456. doi:10.1097/ACI.0000000000000100
8. Abelson MB, Shetty S, Korchak M, Butrus SI, Smith LM. Advances in pharmacotherapy for allergic conjunctivitis. Expert Opin Pharmacother. 2015;16(8):1219-1231. doi:10.1517/14656566.2015.1040760





















