Article
Shifting the Paradigm of Thyroid Eye Disease (TED) Treatment
Author(s):
It is never easy to tell patients that their condition has an uncertain trajectory and treatment options are limited. Until about two years ago, this is what physicians had to tell those living with Thyroid Eye Disease (TED) – a chronic, progressive autoimmune disorder that can cause painful, debilitating, and disfiguring symptoms that may prompt many patients to stop doing the things that they love and withdraw from society.1
I have seen firsthand the impact that TED can have on my patients. In addition to the physical manifestations of TED, it is common for patients to experience psychological distress caused by changes in their appearance. Many tell me they have reduced self-confidence and experience feelings of stress and anxiety. Additionally, these emotions extend beyond the patient and can affect patients’ interpersonal relationships with family, friends, and colleagues.2,3,4
As eyecare providers, TED patients are referred to us with varying eye symptoms. This potentially vision-threatening disease often presents with proptosis, diplopia, pain, and pressure behind the eyes. But more subtle symptoms like eyelid retraction, dryness or grittiness, redness, swelling, and excessive tearing can also be signs of TED.1 Keep in mind, TED is a heterogeneous condition and symptoms can be different from patient to patient. This contributes to the difficulty of diagnosis - oftentimes patients are told for years that they have recurrent pink eye, dry eye, or allergies, resulting in a delay in proper care.5,6
In addition to symptomatic management, it is important to assess the orbit and monitor for optic nerve compression through color vision testing, optic nerve thickness measurements, and visual field testing. Orbital imaging may be indicated to assess the degree of extraocular muscle enlargement and to detect the presence of posterior pole tenting associated with exophthalmos.
Historically, TED had been a “watch and wait” disease because, other than symptom management, not much could be done until patients were candidates for surgery. The waiting period could span years, during which high dose steroids and lubricating eye drops were prescribed to help ease some of the symptoms. When patients are finally candidates for surgery, they undergo sequential procedures addressing exophthalmos, ocular alignment, and eyelid position. In addition to the potential for serious surgical complications (such as CSF leak in orbital decompression surgery),5,7,8 patients also may experience a negative impact on daily life and increased financial burden,1 especially because the procedures require downtime.
The story changed in January 2020 when the U.S. Food and Drug Administration (FDA) approved TEPEZZA® (teprotumumab-trbw) as the first and only medicine for TED. TEPEZZA is an infused biologic, designed to work by inhibiting IGF-1R, a key driver of TED. In doing so, it changes the orbital fibroblast activity, that may result in anatomic changes to the structures behind the eye.9,10 The most common adverse reactions (incidence ≥5% and greater than placebo) are muscle spasm, nausea, alopecia, diarrhea, fatigue, hyperglycemia, hearing impairment, dysgeusia, headache, dry skin, weight decreased, nail disorders, and menstrual disorders.10
Many of my patients have found TEPEZZA to be a good option if they desire a non-steroidal and non-surgical approach that addresses exophthalmos, diplopia, and periocular inflammation concurrently. If the patient is a good candidate for TEPEZZA, I explain that it is an infused medicine administered once every three weeks for a total of eight infusions. These infusions are typically performed at a local infusion center and in some instances, patients can even be infused at home.
One of my patients who saw success with TEPEZZA was a middle-aged female who had double vision and pain associated with TED. She was unable to drive or use a computer, which severely impacted her ability to work and function at home. She felt defeated when surgery was not an option due to the current state of her disease but was interested in trying TEPEZZA. After all eight of her TEPEZZA infusions, her double vision and inflammation resolved. She subsequently underwent only an upper eyelid retraction repair (as opposed to orbital decompression, strabismus surgery, and then eyelid surgery) to restore her appearance.
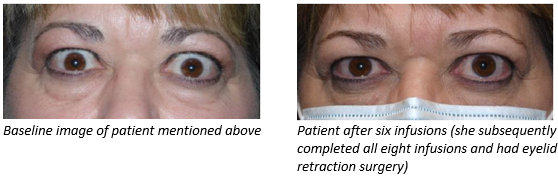
I have also prescribed TEPEZZA in patients who have previously undergone surgeries. For example, an older patient of mine who had lived with TED for several years and had orbital decompression and eyelid surgeries, saw a reactivation in her TED detected by visual field loss. Orbital imaging showed signs of new extraocular muscle expansion and active inflammation. We decided TEPEZZA was the right option for her and saw improved results.
TEPEZZA has been a valuable treatment option to help me manage TED adult patients of various ages. In the elderly population, many live with medical contraindications to high dose steroids or are at higher anesthesia risk for orbital decompression surgery.8
I have found that TEPEZZA is generally well-tolerated, with muscle spasms and nausea being the most frequently reported side effects of my patients. Other side effects were reported in clinical trials and during post-marketing use. I monitor blood glucose levels and collaborate with the patient’s primary care providers and/or endocrinologists to help prevent hyperglycemia. It can also be helpful to partner with an audiologist to conduct baseline and interval hearing exams throughout treatment with TEPEZZA. A small number of patients in clinical trials reported changes in their hearing, like ringing and a sensation of fullness in their ears, that typically resolved within six months after finishing their last infusion.9
With an advancement like TEPEZZA, which treats TED at the source, physicians can now confidently tell their patients that there is more they can do than “watch and wait.” Through a series of outpatient infusions, patients and their doctors can simultaneously address proptosis, diplopia, and periocular inflammation and as a non-steroidal and relatively non-invasive treatment option, it has been a welcome addition to the TED treatment armamentarium.9,10,11,12 It is up to each of us to be aware of the signs and symptoms of TED, spot them early in our patients, and as needed, direct them to a TED Specialist to confirm the TED diagnosis and initiate treatment.
Learn more at TEPEZZAhcp.com.
Newsletter
Don’t miss out—get Ophthalmology Times updates on the latest clinical advancements and expert interviews, straight to your inbox.
