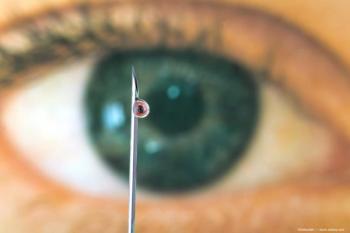
Results favorable for trabecular micro-bypass stent
A trabecular micro-bypass stent is being evaluated for the treatment of refractory open-angle glaucoma in a prospective, multicenter study.
Key Points
New Orleans-A trabecular micro-bypass stent (iStent, Glaukos) appears to be a well-tolerated, safe, and effective modality for lowering IOP in eyes with open-angle glaucoma refractory to prior medical therapy and/or surgery, according to Carlo E. Traverso, MD, speaking here at the annual meeting of the American Academy of Ophthalmology.
Dr. Traverso presented the results from 12 months of follow-up in a prospective, open-label, multicenter study. Researchers enrolled 45 patients with uncontrolled open-angle glaucoma for whom previous conventional surgery-either incisional or laser-had failed; patients were considered to have a poor prognosis for filtration surgery. Efficacy analyses based on patients with successful implantation and those who did not have progression to further surgery showed that implantation of the stent resulted in statistically significant reductions in both mean IOP and average use of IOP-lowering medications. The safety data showed that most complications were related to the surgical procedure. Notably, events traditionally associated with filtering surgery and drainage tubes were absent, and the eyes were strikingly quiet.
"This implant is highly biocompatible, and in postoperative evaluation, it is difficult to determine which eye was operated on," said Dr. Traverso, associate professor, University Eye Clinic, and consulting staff and head of glaucoma and cornea clinical unit, University of Genova Medical School, Genova, Italy. "The results in this challenging group of patients are promising. Now, we need more data from more clinical studies including larger sample sizes and longer follow-up."
The technique involves creation of a stab incision, viscoelastic injection, and positioning of a surgical gonioscopy lens. Then, the introducer, with the implant at the tip, is slid across the anterior chamber. The tip of the implant is engaged into the meshwork, and, once positioned, the implant is released from the introducer with the touch of a button. The surgeon verifies that the device is secure by gently tapping over the implant. Observation of a small amount of blood reflux is a good indication of proper positioning, Dr. Traverso said.
He noted that the implantation technique requires good goniotomy surgery skills.
"I have been doing goniotomy for many years, so I am not the best person to speak about the learning curve for this procedure," he added. "The procedure requires use of a two-handed technique, but I think it certainly is within the reach of the average ophthalmic surgeon."
Enrollment criteria
The enrollment criteria for the prospective study required that patients have best-corrected visual acuity of at least 20/200 and good gonioscopic visibility of the scleral spur. Excluded patients included those with elevated episcleral venous pressure or previous surgery, or conditions where the nasal meshwork was occupied.
All existing IOP-lowering medications were stopped only on the day of surgery, and they were not resumed unless a need arose for further IOP control. Of the patients enrolled, 60% had prior incisional filtration surgery and 30% had previous laser surgery.
The implant could not be implanted in two of the 45 patients enrolled. For the remaining patients, mean IOP was almost 26 mm Hg at baseline and was decreased to just under 19 mm Hg at last follow-up.
"The range of IOP decrease was highly variable-it ranged from just 5 mm Hg in some patients up to 12 mm Hg," noted Dr. Traverso.
Outcomes
Considering the patients who had not undergone further surgery, mean IOP decreased 25% from baseline at 12 months. Use of IOP-lowering medications was reduced from a mean of 2.1 at baseline to about one. As expected, however, a trend was seen whereby use of IOP medications increased over time.
Complete success, defined as IOP of 21 mm Hg or less without need for any glaucoma medications, was achieved in 21% of patients, and qualified success-defined as the same IOP level with or without medication use-was thought to have been achieved in 79% of patients.
Adverse events included partial obstruction of the stent by the iris in two patients, both being successfully treated. Procedure-related adverse events included four iris touches with no subsequent damage, one iris root touch with bleeding, two intraoperative anterior chamber collapses, and three cases of excessive bleeding.
Newsletter
Don’t miss out—get Ophthalmology Times updates on the latest clinical advancements and expert interviews, straight to your inbox.