Article
News
Meeting the need: Screening parameters for presbyopia-correcting drops
Author(s):
Ophthalmologists should consider a few key points before writing a prescription.
(Image courtesy of Adobe Stock)
Donna L. Shotwell, OD, also contributed to this article.
When presbyopia-correcting drops were introduced, many eye care providers thought they would be a quick, easy fix for the unhappy presbyopes in our offices, perhaps envisioning wide distribution of samples and on-demand prescribing. That was probably never the best approach, especially when non–eye care practitioners started prescribing the medication.
Late 2022 saw the publication of several case reports1-3 of patients who experienced retinal detachment (RD), retinal tears, or vitreomacular traction following the use of topical pilocarpine 1.25% (Vuity; Allergan/AbbVie) for presbyopia correction. The American Society of Cataract and Refractive Surgery convened a task force and issued recommendations.4 Allergan/AbbVie updated the warnings and precautions section of Vuity’s labeling to clarify the risk of RD, especially in patients with prior retinal problems.
Still, the risk of RD is small, especially in appropriately selected patients. We encourage colleagues to mirror closely the patient population studied in clinical trials when prescribing presbyopia-correcting drops, at least initially. There have not been any cases of RD or other serious complications among clinical trial participants, probably because they were low risk to begin with. For the millions of presbyopic patients who are dependent on having reading glasses stashed all over the house, a topical pupil-modulating drop is a convenient, effective approach for improving near vision. This article will discuss screening measures we find essential for ensuring the safe, successful use of presbyopia-correcting drops.
Minimize risk
It is prudent to perform a thorough eye examination, including a dilated view of the peripheral retina, before prescribing topical pilocarpine 1.25% or any other presbyopia-correcting eye drop. One should look carefully for areas of lattice degeneration and retinal thinning that could be prone to tearing with ciliary body contraction induced by the miotic agent. In some cases, it may be desirable to obtain optical coherence tomography of the macula to discern whether there is evidence of previous or evolving posterior vitreous detachment (PVD) or vitreomacular traction.
A history of retinal problems, retinal laser surgery, or high myopia should also be considered risk factors for RD. We have many postrefractive and pseudophakic patients in our practices who are now emmetropic but are interested in better near vision. If they were high myopes before refractive surgery, they still have the anatomy of a high myope and probably should avoid pilocarpine. Conversely, one might feel less concerned about a patient who has already had a PVD, because there is no longer as much traction on the retina.
The Gemini 1 (NCT03804268) and 2 (NCT03857542) and Virgo (NCT04983589) clinical trials excluded high myopes—indeed, everyone outside a narrow refractive range of –4.00 diopters (D) to +1.00 D—as well as pseudophakes and patients with any history of retinal pathology. As noted, in these clinical trials, there were no cases of RD. At least half of the 6 cases of RD reported in the literature outside of these clinical trials had significant risk factors, and the rest may or may not have had a retinal exam prior to prescribing.
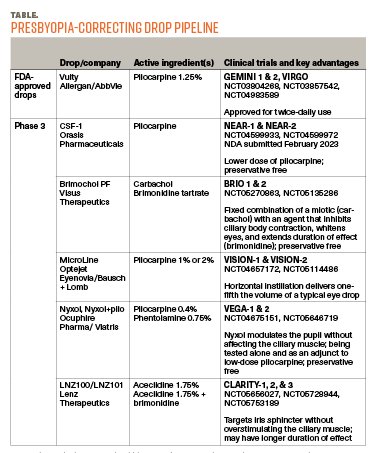
At present, we have 1 presbyopia-correcting drop to offer patients, but at least 5 other companies’ offerings—some of which may offer a lower risk of RD—are making their way through the clinical trial and regulatory process (Table). Pilocarpine products from Ocuphire and Orasis, for example, are expected to contain lower doses of pilocarpine that may be less likely to provoke RD. Aceclidine, the active ingredient in LNZ100 (LENZ Therapeutics), may have less impact on ciliary body contraction, although laboratory studies in cadaver and bovine eyes have produced mixed results.5,6 The brimonidine in Brimochol PF (Visus Therapeutics) has also been shown to inhibit ciliary body contraction in bovine eyes in the laboratory.7
Maximize the chance of success
Safety is not the only reason to be discriminating about which patients should be offered presbyopia-correcting drops. Patients’ refractive status, degree of residual accommodation, and visual needs will also affect how well these drops work for them.
Latent hyperopes likely will not see much near benefit from presbyopia-correcting drops, although they may get a distance benefit from a smaller pupil. Similarly, a –6.0 D myope aged 55 years with very little remaining natural accommodation also may not see sufficient effect from the current presbyopia-correcting drops.
The perfect candidate may be an early presbyope who has never worn glasses or contact lenses. These patients may be too young to consider lens surgery and typically do not do well in multifocal contact lenses because the lenses are unlikely to provide the quality of vision to which they are accustomed.
Expectations also matter. A J10 patient is simply not going to get to J1 or J2—but perhaps that patient would still benefit from better computer vision when using the drops. It is critical to ask patients how they are reading now and in which situations they would like to function better. With pilocarpine 1.25%, patients will have to use it twice daily (as was recently approved) to extend the effects to a full work day. We can expect that future presbyopia-correcting drops may have a longer duration of effect with a single instillation, and perhaps a higher efficacy rate—that is, a higher percentage of patients who achieve the approval without any loss of distance acuity. For example, Visus recently announced that its drop, Brimochol PF, achieved highly statistically significant near vision improvements over 8 hours.
Among the lessons we have learned with Vuity is that using a presbyopia-correcting drop for 1 day is not sufficient to judge its performance or adverse effects. The Virgo study that led to the twice-daily indication for Vuity showed an increase in efficacy over time: Patients had better results at 14 days than at 7 and better results at 7 days than on the first day. Additionally, transient hyperemia or headache is common on the first instillation but diminishes. For these reasons, we typically recommend that patients try the drops for a week before deciding whether they see a benefit.
Although it is important to screen patients carefully and educate them about the risks and potential adverse effects of presbyopia-correcting drops, it is still beneficial to have something as easily reversible as a topical drop for a population that has few other good options for correcting presbyopia. As more presbyopia-correcting drops are approved, the benefits we can offer patients will continue to grow and expand, as well.
Cathleen M. McCabe, MD
E: cmccabe13@hotmail.com
McCabe is chief medical officer at Eye Health America and medical director at The Eye Associates in Bradenton, Florida She is a consultant for Visus and AbbVie and has received research support from Orasis and Allergan.
Donna L. Shotwell, OD
E: drdonnashotwell@gmail.com
Shotwell is a practice owner and optometrist in Sarasota, Florida. She has no financial relationships to disclose related to this article.
References:
1. Al-Khersan H, Flynn HW Jr, Townsend JH. Retinal detachments associated with topical pilocarpine use for presbyopia. Am J Ophthalmol. 2022;242:52-55. doi:10.1016/j.ajo.2022.05.011
2. Eton EA, Zhao PY, Johnson MW, Rao RC, Huvard MJ. Rhegmatogenous retinal detachment following initiation of pilocarpine hydrochloride ophthalmic solution 1.25% for treatment of presbyopia. Retin Cases Brief Rep. Published online August 12, 2022. doi:10.1097/ICB.0000000000001309
3. Amarikwa L, Michalak SM, Caul S, Mruthyunjaya P, Rahimy E. Vitreofoveal traction associated with pilocarpine for presbyopia. Ophthalmic Surg Lasers Imaging Retina. 2022;53(7):410-411. doi:10.3928/23258160-20220629-01
4. Rhee D, McCabe C, Herndon L, Rao R. ASCRS consensus statement on VUITY. American Society of Cataract and Refractive Surgery. October 2022. Accessed June 20, 2023. https://ascrs.org/clinical-education/clinical-reports/2022-ascrs-consensus-statement-on-vuity
5. Ishikawa H, DeSantis L, Patil PN. Selectivity of muscarinic agonists including (+/–)– aceclidine and antimuscarinics on the human intraocular muscles, J Ocul Pharmacol Ther. 1998;14(4):363-373. doi:10.1089/jop.1998.14.363
6. Lepple-Wienhues A, Stahl F, Wiederholt M. Differential smooth muscle-like contractile properties of trabecular meshwork and ciliary muscle. Exp Eye Res. 1991;53(1):33-38. doi:10.1016/0014-4835(91)90141-z
7. Kubo C, Suzuki R. Involvement of prejunctional alpha 2-adrenoceptor in bovine ciliary muscle movement. J Ocul Pharmacol. 1992;8(3):225‑231. doi:10.1089/jop.1992.8.225
Newsletter
Don’t miss out—get Ophthalmology Times updates on the latest clinical advancements and expert interviews, straight to your inbox.





