CorNeat Vision’s scleral patch, EverPatch, hits the US market
The patch is the world's first non-degradable, synthetic tissue substitute for ophthalmic surgery.
(image Credit: AdobeStock/konoplizkaya)

CorNeat Vision announced the CorNeat EverPatch, the first synthetic and non-degradable tissue substitute for use in ophthalmic surgeries is available on the US market after receiving 501(k) clearance from the US Food & Drug Administration (FDA) back in June 2023.
According to CorNeat,1 the EverPatch is designed to “revolutionize ophthalmic surgeries” and has a “unique combination of features that provide a significant advantage over existing tissue grafts, which degrade over time.” It is composed of a non-woven, polymer matrix, which integrates with surrounding tissue and is intended to reinforce the sclera and aid the physical reconstruction of the ocular surface.
The device is sized to be used and as such, should not be trimmed; measuring 0.5 cm x 0.65 cm with 100 microns thickness, which according to the company allows it to better "conform to the eye wall.”2,3
Gilad Litvin, MD, Co-Founder and Chief Medical Officer at CorNeat Vision, emphasizes the safety and efficacy of the CorNeat EverPatch in a press release from the company.
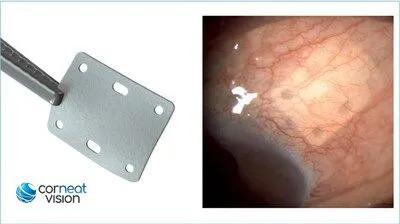
left - CorNeat EverPatch; right - CorNeat EverPatch implanted under the conjunctiva, 9 months post implantation. (Photo Courtesy CorNeat Vision)
"Our synthetic tissue substitute is tear-resistant, non-degradable, and completely bio-compatible, with no observed chronic inflammatory response that exists around tissue grafts. We are tracking over one hundred devices that we supplied to 25 US ophthalmic centers in the past couple of months, with excellent feedback. We are confident that the CorNeat EverPatch will set a new standard of care in ocular surgeries necessitating scleral reinforcement,” said Litvin.
According to the company, it plans to showcase the EverPatch as the upcoming American Glaucoma Society (AGS) and American Society of Cataract and Refractive Surgery (ASCRS) Annual Meetings.1
Outside of the US, the company aims to obtain CE marketing in early 2025.2
References:
CorNeat EverPatch Hits the US Market: Farewell to Traditional Ocular Tissue Grafts. Press release. Accessed February 27, 2024. Published February 27, 2024. https://www.prnewswire.com/il/news-releases/corneat-everpatch-hits-the-us-market-farewell-to-traditional-ocular-tissue-grafts-302071176.html
510(K) Summary of CorNeat EverPatch. Accessed February 27, 2024. https://www.corneat.com/_files/ugd/cd910f_adba70b6560f43e98f883d2c7f562cc0.pdf
CorNeat EverPatch. Accessed February 27, 2024. https://www.corneat.com/corneat-everpatch-us