|Videos|September 1, 2020
Combined MIGS & Cataract Surgery Case Study with Dr. Paul Singh
Author(s)SPONSORED BY BAUSCH + LOMB
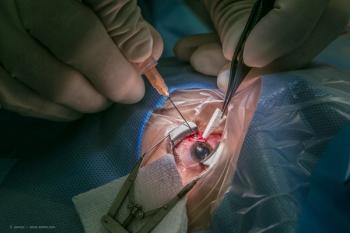
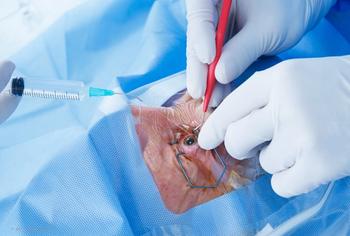
Dr. Paul Singh manages postoperative inflammation and pain with LOTEMAX® SM following combined MIGS & Cataract surgery. Interested in learning more?
Advertisement
Sponsored by Bausch + Lomb
Paul Singh, MD
The Eye Centers of Racine & Kenosha
Dr. Paul Singh manages post-operative inflammation and pain with
Interested in learning more? Visit
Advertisement
Latest CME
Advertisement
Advertisement
Trending on Ophthalmology Times - Clinical Insights for Eye Specialists
1
TearSolutions advances Lacripep into phase 2 following dual FDA designations in neurotrophic keratitis
2
Oculis phase 3 DIAMOND trials miss vision endpoints in diabetic macular edema
3
Opuviz aflibercept biosimilar launches in Europe for retinal diseases
4
Study links alcohol consumption to earlier advanced presbyopia in men
5