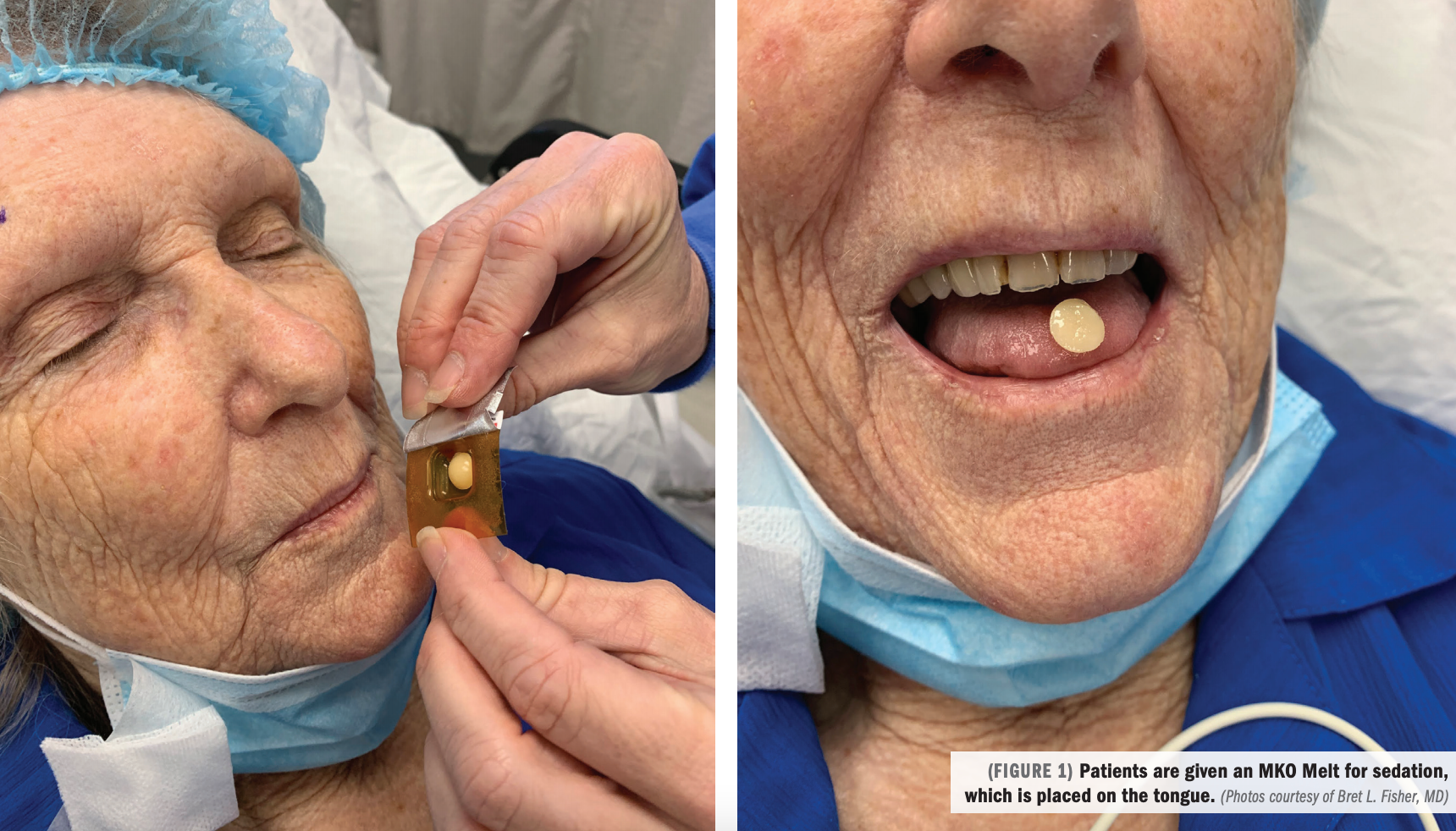
Pharmacy
Latest News
Latest Videos

CME Content
More News

Investigators looked at trends by geographic variation in practice patterns, perceptions toward opioids, and patient population

This action will result in 2 separate companies,

Bepostastine besilate ophthalmic solution 1.5% (Bepreve, ISTA Pharmaceuticals) is safe and well-tolerated in healthy children as young as 3 years of age, according to results from a multicenter, prospective, double-masked, randomized, placebo-controlled trial.

Blink Tears Lubricating Eye Drops improved dry eye symptoms significantly better than Systane Lubricant Eye Drops when the artificial tears were paired with topical cyclosporine ophthalmic emulsion.

For patients who are suffering with moderate to severe signs and symptoms of seasonal or perennial allergic conjunctivitis, loteprednol etabonate ophthalmic suspension 0.2% (Alrex, Bausch & Lomb) is a safe and well-tolerated therapeutic option that can provide rapid clinical improvement, according to new research.

An efficacy evaluation comparing two marketed artificial tears showed that a polyethylene-based product (Systane Ultra, Alcon Laboratories) resulted in statisically greater reductions in corneal and conjunctival staining than a glycerin/carboxymethylcellulose-based tear (Refresh Optive, Allergan).

The overall anti-allergic eye drop market consists of about 25 products developed and approved in the United States (with some marketed abroad) to address the needs of the seasonal allergic conjunctivitis patient population.

The use of a hydroxypropyl cellulose ophthalmic insert (Lacrisert, Aton Pharma) has been proven to offer much relief to patients suffering from dry eye syndrome.

A phase II study with a novel non-invasive iontopohoretic drug delivery system (EyeGate II Delivery System, EyeGate Pharma) shows that the device appears to optimize the delivery of a proprietary dexamethasone-derived corticosteroid solution (EGP-437) and improve significantly the signs and symptoms of dry eye, which may raise the bar in therapies for dry eye syndrome.

Results of a utility analysis of cyclosporine 0.05% ophthalmic emulsion (Restasis, Allergan) show that it is highly cost-effective and improves the quality of life in patients with moderate to severe dry eye disease that is unresponsive to conventional lubricant therapy.

Analyses of data from two hybrid environmental studies evaluating olopatadine hydrochloride 0.2% once daily for the treatment of seasonal allergic conjunctivitis or rhinoconjunctivitis show that patients derive improvement in the frequency and/or severity of their nasal symptoms as a simultaneous benefit with relief of ocular allergy, according to Mark B. Abelson, MD.

A new eyelid spray (Tears Again advanced liposome spray, OcuSOFT) is available that may benefit patients suffering from meibomian gland dysfunction.

Given the plethora of over-the-counter and prescription approaches available for dry eye, patient and clinician agreement on the appropriate regimen is key to treatment success.

The multimodal anti-allergy agent epinastine hydrochloride 0.05% (Elestat, Inspire Pharmaceuticals) is an ideal treatment option for the management of seasonal allergic conjunctivitis and is a particularly good choice in patients who have dry eye disease as a comorbidity, according to Laurie D. Barber, MD

Grid laser photocoagulation remains standard of care for eyes with ME secondary to BRVO.

Many approaches for treating exudative age-related macular degeneration (AMD) have been developed.

An updated analysis of data from an ongoing phase II study of intravitreal ranibizumab treatment for diabetic macular edema shows that functional and anatomic benefits observed at 6-month primary efficacy endpoint are generally maintained at 1 year after switching to a "prn" dosing schedule.

A preliminary study of epimacular brachytherapy showed that radiation and antivascular endothelial growth factor injections reduced the treatment burden in patients with neovascular age-related macular degeneration.

Alcon Laboratories' Independent Director Committee is calling Novartis' merger proposal "grossly inadequate," and it is vowing to fight Novartis in its efforts to take control.

A single sustained-release dexamethasone intravitreal implant resulted in resolution of inflammation and improvement in visual acuity (VA) in patients with intermediate or posterior uveitis through 26 weeks in a multicenter, randomized, controlled clinical trial.

Pooled results from two randomized, investigator-masked parallel group clinical trials show that the fixed combination fo brimonidine tartrate 0.2%-timolol maleate 0.5% produced greater IOP lowering than the fixed combination of dorzolamidehydrochloride 2%-timolol maleate 0.5%, whether the fixed combination products were used alone or as an adjunct to treatment with a prostaglandin analogue.

Timolol maleate 0.5%/sorbitol complex is a convenient, well-tolerated choice for adjunctive therapy in patients taking a prostaglandin analogue who need further IOP control.

Switching to another prostaglandin analogue that does not contain benzalkonium chloride may not be the answer for managing complaints from patients of dryness and irritation.

Novartis plans to assume complete control over Alcon Laboratories in a two-step process that some analysts and some members of Alcon's board of directors are challenging for its fairness under U.S. and Swiss merger laws.

Glaucoma and ocular hypertension can effectively be treated topically with IOP-lowering drops.